Mild Cognitive Impairment (MCI) Clinical Trial Recruitment | University of Toronto | Unity Health Toronto | Vielight Neuro (n=60)
Do you—or someone you love—notice recent memory changes? You may be eligible for a research study at St. Michael’s Hospital in Toronto. This study investigates PBM therapy, a non-invasive method that uses near-infrared (NIR) light—to see whether it can support brain health in people with memory concerns.
For more details about this MCI clinical trial: https://clinicaltrials.gov/study/NCT06618807
Why this study matters
MCI is a transitional state between normal aging and Alzheimer’s dementia. On average, about 32% of people with MCI progress to dementia, 62% remain stable, and roughly 6% return to normal cognition at later visits. While some medications (e.g., donepezil, galantamine, rivastigmine, memantine) can help with symptoms, there’s still a need for safe options that may support longer-term cognitive function with fewer side effects.
This study investigates PBM therapy—a non-invasive method that uses gentle near-infrared (NIR) light—to see whether it can support brain health in people with memory concerns.
About the study
-
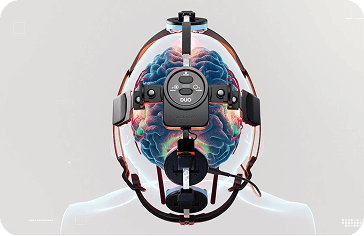
Device tested: Neuro RX Gamma, a wearable headset with 5 LEDs plus an intranasal applicator (1 LED).
-
How the device works: The Vielight Neuro delivers 810nm NIR light to targeted brain regions. It is non-thermal (does not produce significant heat) and non-invasive.
-
Design: 60+ participants will use either an active or sham (placebo-like) Neuro RX Gamma device, allowing researchers to compare outcomes fairly.
-
Duration: 12 weeks total.
-
Location: Toronto.
Important: PBM is being studied here to understand potential benefits; it is not an approved treatment for MCI. Participation is voluntary.
Who can apply
You may be a good fit if you:
-
Are 50 years or older, and
-
Have noticed memory changes (e.g., forgetfulness, word-finding, misplacing items), without a formal diagnosis of MCI or dementia.
- You are able to undergo a blood test and an MRI scan (Exceptions for undergoing MRI may be allowed)
If you’re unsure, the study team can guide you through a brief screening to check eligibility.
What participation looks like
- Screening & consent: Undergo an initial assessment to determine eligibility for the study.
- Baseline visit: Assessments of memory and thinking, blood draw, and brain MRI (MRI exceptions allowed)
- At-home use: 20 minutes daily, 6 days a week, for 8 weeks
- Check-ins: Short follow-ups to track how you’re doing.
- Post-Trial Evaluation: Repeat assessments to measure any changes over the 8 weeks.
- Follow-up visit: 4 weeks after the Post-Trial Evaluation, repeat memory assessments and blood draw to measure lasting effects.
The team will teach you exactly how to place and use the device. LEDs are standard, semiconductor light sources—no surgery, no needles.
Safety
PBM at low levels is generally considered safe and well-tolerated in research settings. As with any study, you’ll be informed of potential risks and can withdraw at any time.
Ready to learn more or sign up?
Phone: 416-360-4000 ext. 47838
Email: memoryclinic@unityhealth.to
When you reach out, you can simply say:
“I’m interested in the photobiomodulation memory study for people over 50.”
Quick FAQ
Is this a treatment?
This is a clinical research study to evaluate PBM; it is not established treatment for MCI.
Do I need a diagnosis first?
No. If you’re over 50 and noticing memory changes—but don’t have a formal diagnosis—you’re welcome to contact the team for screening.
Will I definitely get the active device?
Not necessarily. To ensure fair results, some participants use a sham device. You’ll learn which you had after the study, if applicable.
Can I keep my current medications?
Please tell the study team about any medications. They’ll advise you on eligibility and any required adjustments.
About the Device
The Neuro RX Gamma, a wearable headset with 5 LEDs plus an intranasal applicator (1 LED).
It delivers 810nm NIR light to targeted brain regions. It is non-thermal (does not produce significant heat) and non-invasive.
Safety: PBM at low levels is generally considered safe and well-tolerated in research settings. As with any study, you’ll be informed of potential risks and can withdraw at any time.
Important: PBM is being studied here to understand potential benefits; it is not an approved treatment for MCI. Participation is voluntary.
Share with a friend or family member
If you know someone 50+ who’s been forgetting appointments, repeating questions, or feeling more “foggy” than usual, please share this post. Early participation in research can help advance options for everyone.
This post is for recruitment and general information only. It does not replace medical advice. Speak with your healthcare provider about any concerns regarding your memory or thinking