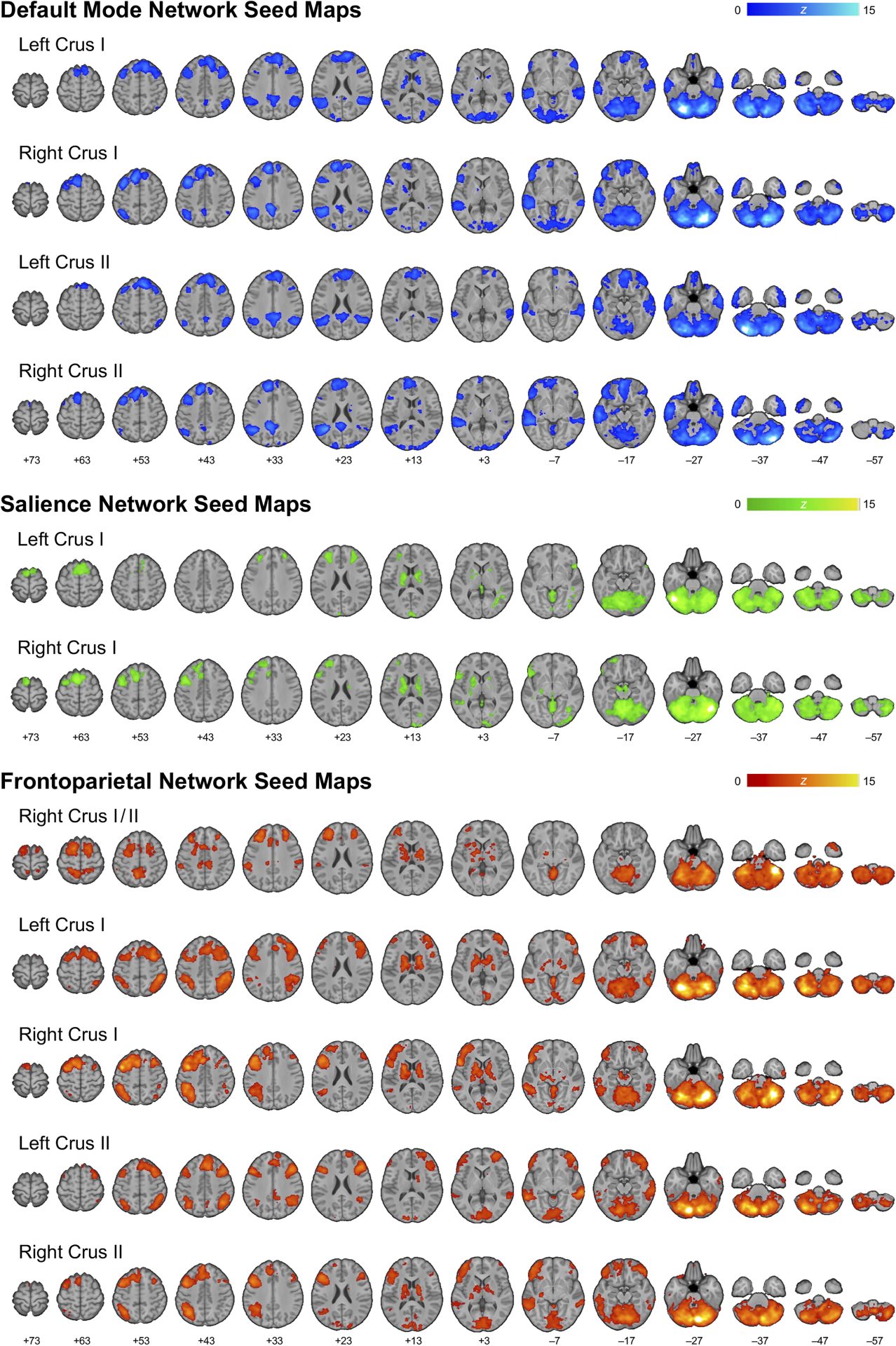
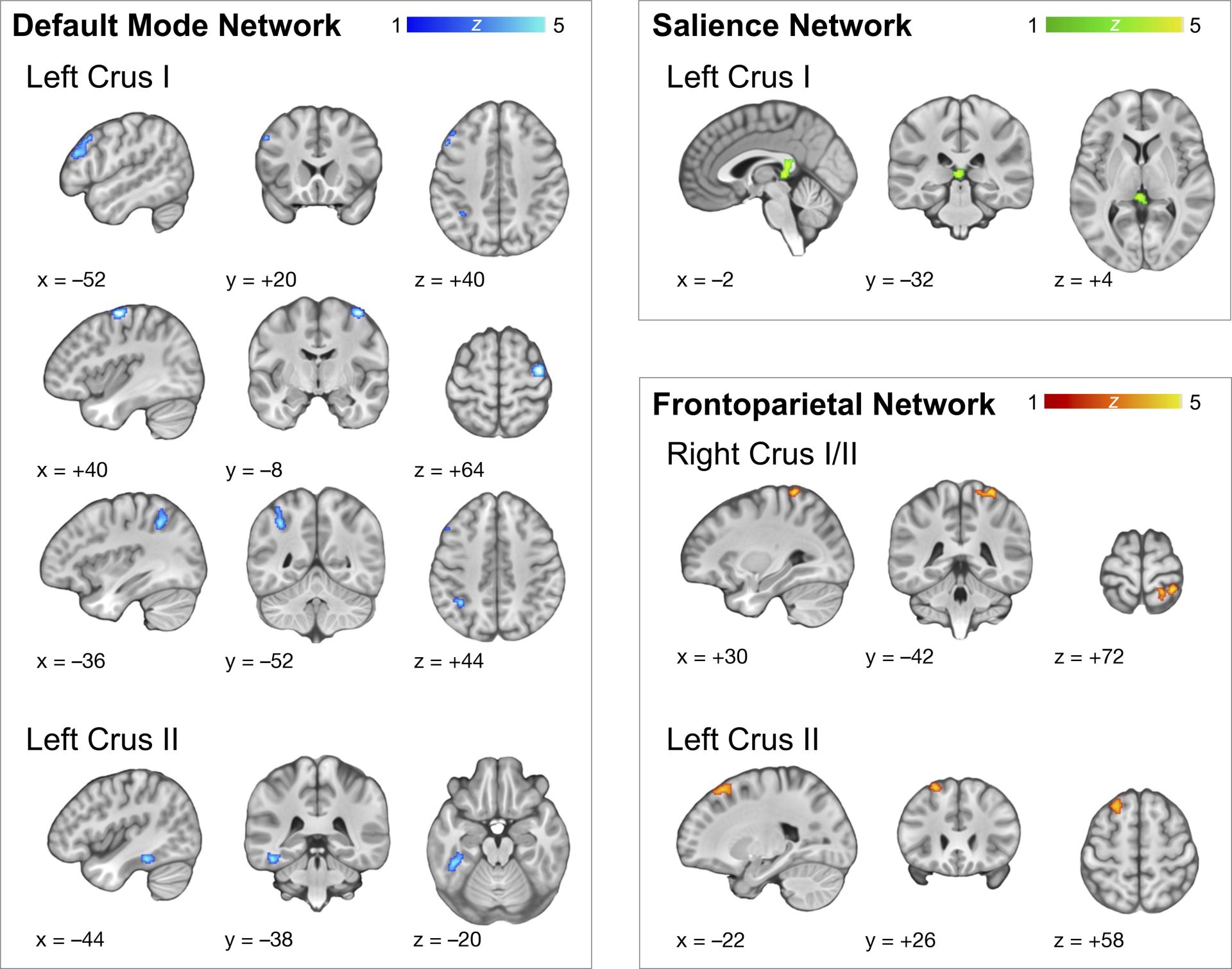
Brain Imaging Studies with Vielight Neuro Reveal the Network Effects of Brain Photobiomodulation
For years, brain photobiomodulation (PBM) was often described in local terms: more blood flow here, less inflammation there, happier mitochondria everywhere.
But over the last few years, something deeper has been coming into focus.
Independent MRI and fMRI studies from the University of Sydney, the University of California San Francisco (UCSF), and the University of Utah are showing that PBM with Vielight technology doesn’t just act locally. It appears to reshape how large-scale brain networks behave – especially the Default Mode Network (DMN) and its partners in attention and cognitive control.
This post walks through three key imaging studies:
- Healthy adults – neuromodulation and DMN deactivation during a task (University of Sydney)
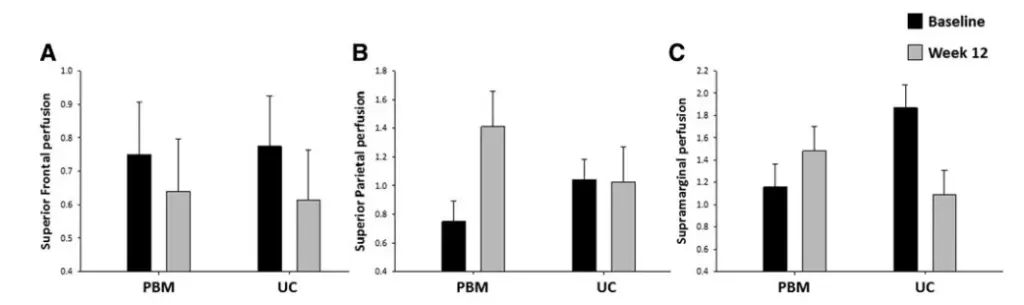
- Dementia/Alzheimer’s – perfusion and DMN connectivity at rest (University of California San Francisco & VA San Francisco)
- Traumatic brain injury – cerebellar network efficiency in ex-football players (University of Utah)
- Mild Cognitive Impairment (MRI) – Pending Publication (Preprint – University of Toronto)
- Traumatic brain injury (DTI) – Pending Publication (University of Utah)
Together, they outline a consistent story: Vielight PBM strengthens the efficiency of core neural networks.
⚠️ These are research findings, not medical advice.
A quick refresher: What is the Default Mode Network?

The Default Mode Network is a set of regions (including the posterior cingulate cortex, medial prefrontal cortex, and lateral parietal areas) that typically:
-
Are more active when the mind is resting, daydreaming, or internally focused, and
-
Should deactivate when we shift into demanding tasks.
In many neurological conditions — including Alzheimer’s disease and TBI — this network shows abnormal connectivity and dysregulated activity, such as reduced within-network coherence or failure to deactivate appropriately during tasks.
That’s why DMN behavior is such a central theme in the PBM imaging story.
The Vielight Neuro 4’s geometry is engineered to intentionally overlap these broadened fields over Default Mode Network (DMN) nodes with the highest measured irradiance in commercially available brain photobiomodulation devices, while still bathing the wider cortex. This is why five VieLED modules can produce an effect that is effectively full‑transcranial, with a focus on the DMN.
Imaging Study 1 (MRI): University of Sydney: DMN deactivation in healthy young adults
Study: (Link)
El Khoury, H., Mitrofanis, J., & Henderson, L. A. (2019).
“Exploring the Effects of Near Infrared Light on Resting and Evoked Brain Activity in Humans Using Magnetic Resonance Imaging” (Neuroscience).
Device & protocol
-
Technology: Vielight Neuro
-
Population: 24 Normal, young adults.
-
Design: Participants underwent active vs sham PBM sessions, then performed fMRI at rest and during a finger-tapping task.


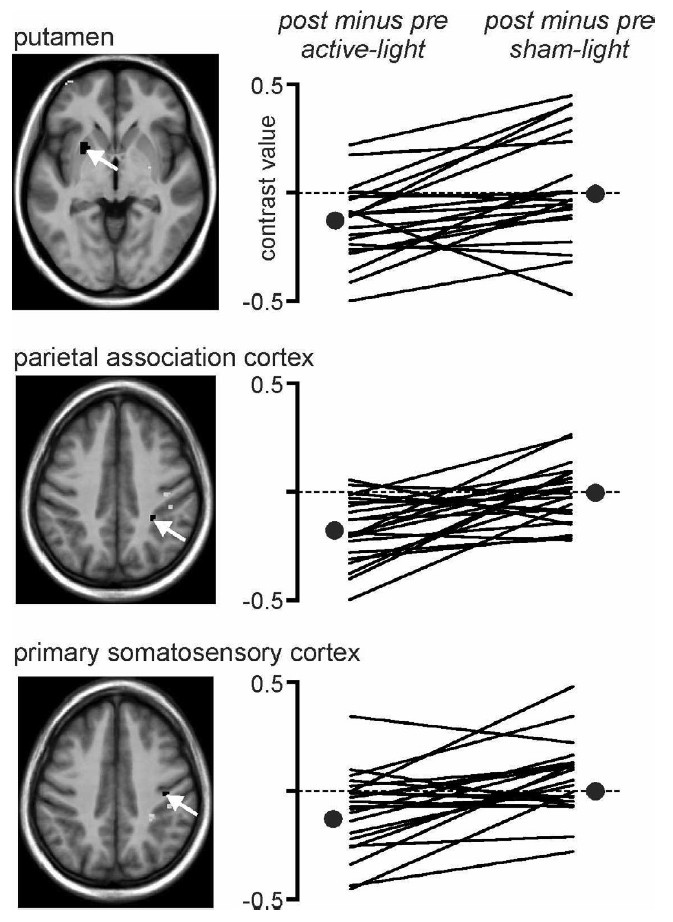
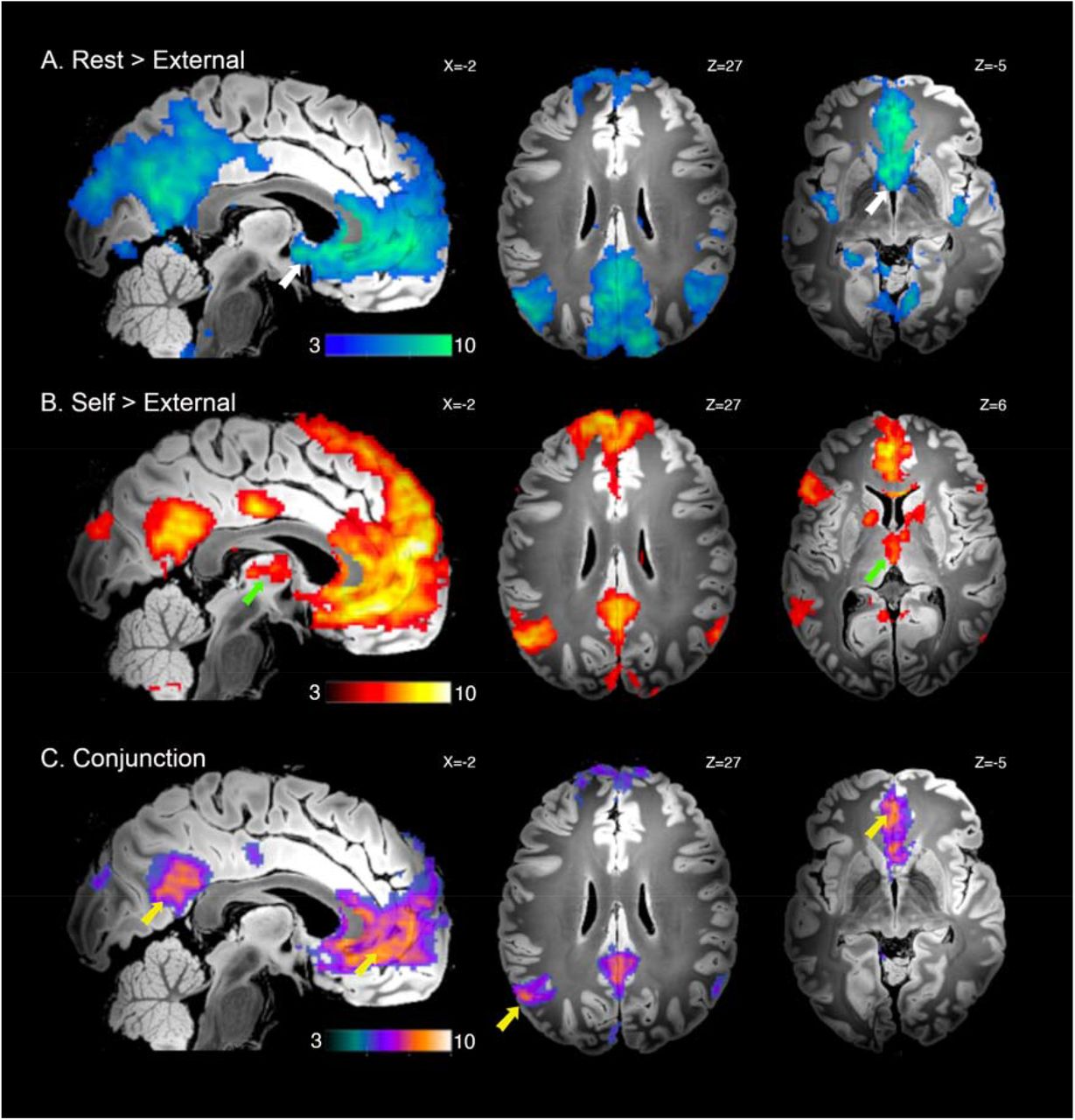
Figure 1. Active vs Sham - Vielight Neuro Effects of active- and sham-light on finger-tapping evoked activity. (A) Significant differences in finger-tapping evoked activation before and after active-light
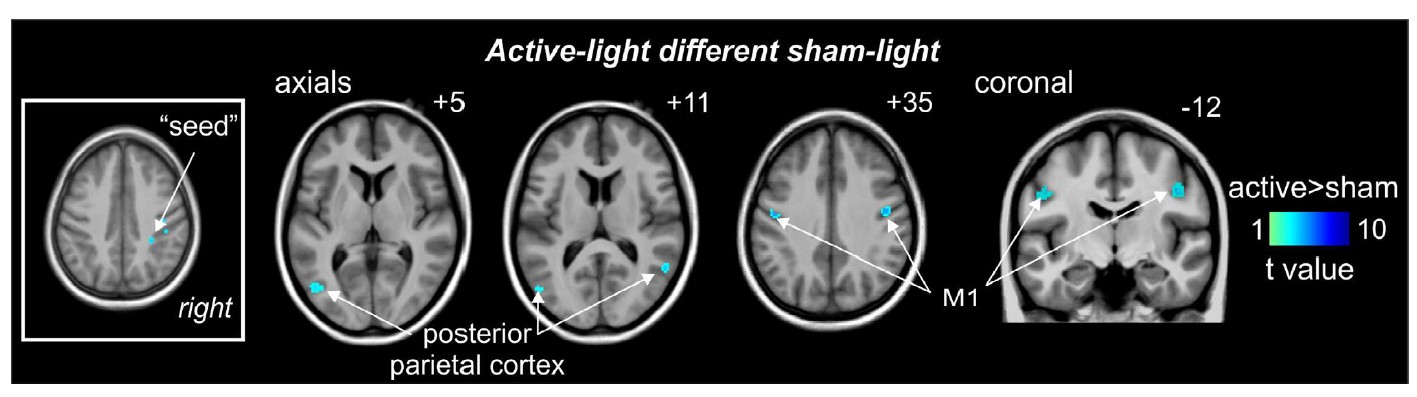
At the University of Sydney, researchers asked an important question: if healthy young adults receive a single session with a Vielight 810 nm transcranial device, what changes on fMRI at rest and during a simple finger-tapping task?
Their answer: resting global activity didn’t budge, but during the task there were reductions in activity across regions you want to quiet when focusing (primary somatosensory cortex, parietal association cortex, putamen). Functional connectivity from parietal association cortex shifted as well. The team interprets these shifts as attention/novelty related and notes they “served to deactivate the so-called default mode network (DMN).”
Why that matters: in a healthy brain, the DMN should dial down when you focus. This study reads as PBM acting like context-sensitive gain control—not revving a resting brain, but helping it suppress background chatter the moment work begins.