Groundbreaking U of Utah TBI Study with Vielight Neuro | The Effect of Intranasal Plus Transcranial Photobiomodulation on TBI
This article is based on independent TBI research conducted with the Vielight Neuro Gamma by the University of Utah
Educational summary of published clinical research on intranasal-transcranial photobiomodulation (itPBM) using Vielight technology in individuals with a history of repetitive head acceleration events (RHAEs) and related cognitive concerns. The Vielight Neuro is categorized as a general wellness device by the FDA. It is not cleared or approved by the U.S. FDA to diagnose, treat, cure, mitigate, prevent, or manage traumatic brain injury (TBI).
Brain photobiomodulation (PBM) with the Vielight Neuro Gamma was explored as a therapy for brain injuries by neurology professors from the University of Utah with Vielight’s patented simultaneous intranasal and transcranial PBM (itPBM) technology.
The Vielight Neuro Gamma utilizes specialized, patented light-emitting diodes (Vie-LEDs) to target brain regions with near-infrared light, promoting energy production, blood flow, and cell survival while reducing inflammation. This therapy may also enhance neuromuscular health by improving reaction time, dexterity, grip strength, and balance.
This study enrolled 49 participants (43 completed) aged 18-69 years, all with self-reported mTBI or RHAEs from sports or other activities. The study aimed to examine the effects of itPBM on neuromuscular health and found potential benefits, suggesting that PBM might improve or preserve physical performance and neuromuscular function.
“Football almost killed me… But Vielight saved my life.” — Dr. Larry Carr.

Study Design
This study was a nonrandomized proof-of-concept design focusing on active treatment only. Participants were assessed before and 8-10 weeks after starting at-home itPBM treatments. They were instructed to maintain their normal routines and avoid new activities such as resistance or brain training.

Figure 1 – Participants Backgrounds (n=43)
Treatment Protocol
Participants self-administered itPBM using the Vielight Neuro Gamma PBM headset, which has four LEDs emitting NIR light (810 nm) and an intranasal probe emitting pulsed NIR light. Treatments lasted 20 minutes every other day for 8 weeks, with participants tracking usage on a log sheet.

Figure 2 – Placement of the Vielight Neuro Gamma over the Default Mode Network and intranasal
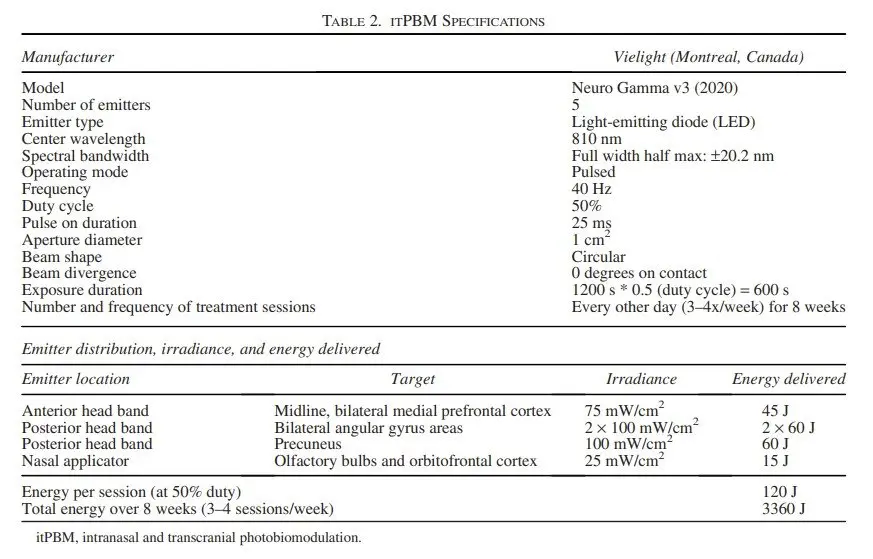
Figure 3 – PBM Parameters of the Vielight Neuro device
Clinical Performance Tests
Four assessments were conducted alongside cognitive and neuropsychological tests and a brain MRI:
- Clinical Reaction Time
Participants caught a falling stick with their dominant hand to measure reaction time, calculated using the distance the stick dropped and the formula for a body falling under gravity. - Grooved Pegboard Test
Manipulative dexterity was assessed using a metal board with 25 keyhole-shaped slots. Participants inserted pegs into the holes as quickly as possible, recording the time taken to complete the task with each hand. - Grip Strength
Measured using a Jamar Plus Dynamometer, participants squeezed the handle as hard as possible for 3 seconds, with three trials per hand. The average force was recorded. - Mini Balance Evaluation Systems Test (MiniBEST)
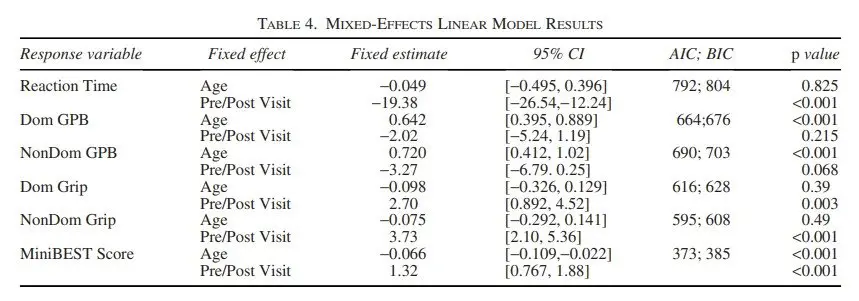
This test assessed 14 tasks across four domains (anticipatory postural adjustments, reactive postural control, sensory orientation, and dynamic gait), scoring each task on a three-level ordinal scale with a maximum total score of 28. The overall score and individual subscores for each domain were recorded.
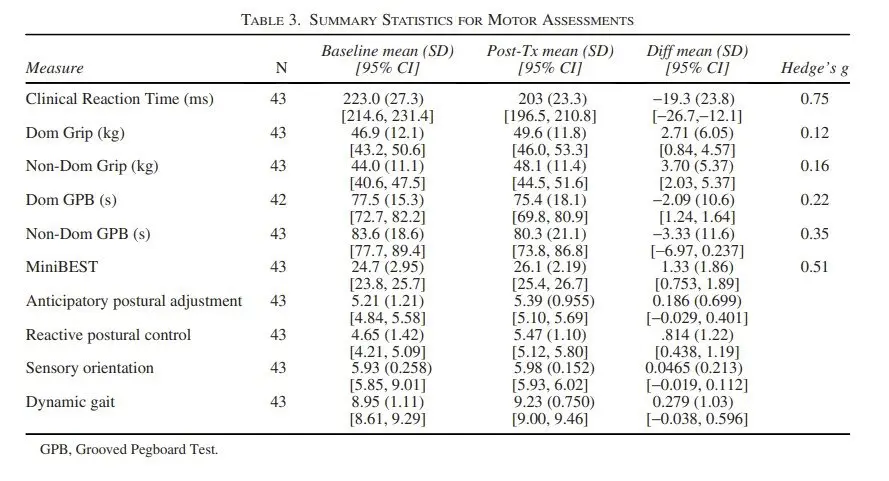
Figure 4 – Summary Statistics of the Motor Assessments
Statistical Analysis
Motor tests were scored according to their standards. Data from participants with limb injuries were excluded. Normality of measurements was evaluated using Shapiro-Wilk tests. Linear mixed-effect analyses, using the maximum likelihood method, assessed motor outcomes with “age” and “visit time” as fixed effects and random intercepts by “participant.” Normality was confirmed through residuals examination. Wilcoxon’s signed-rank test was used for non-normal variables. The Benjamini-Hochberg procedure controlled for multiple comparisons, with a significance threshold of α = 0.05. P values with 95% confidence intervals (CIs) were reported, and effect size was measured by Hedges’ g.